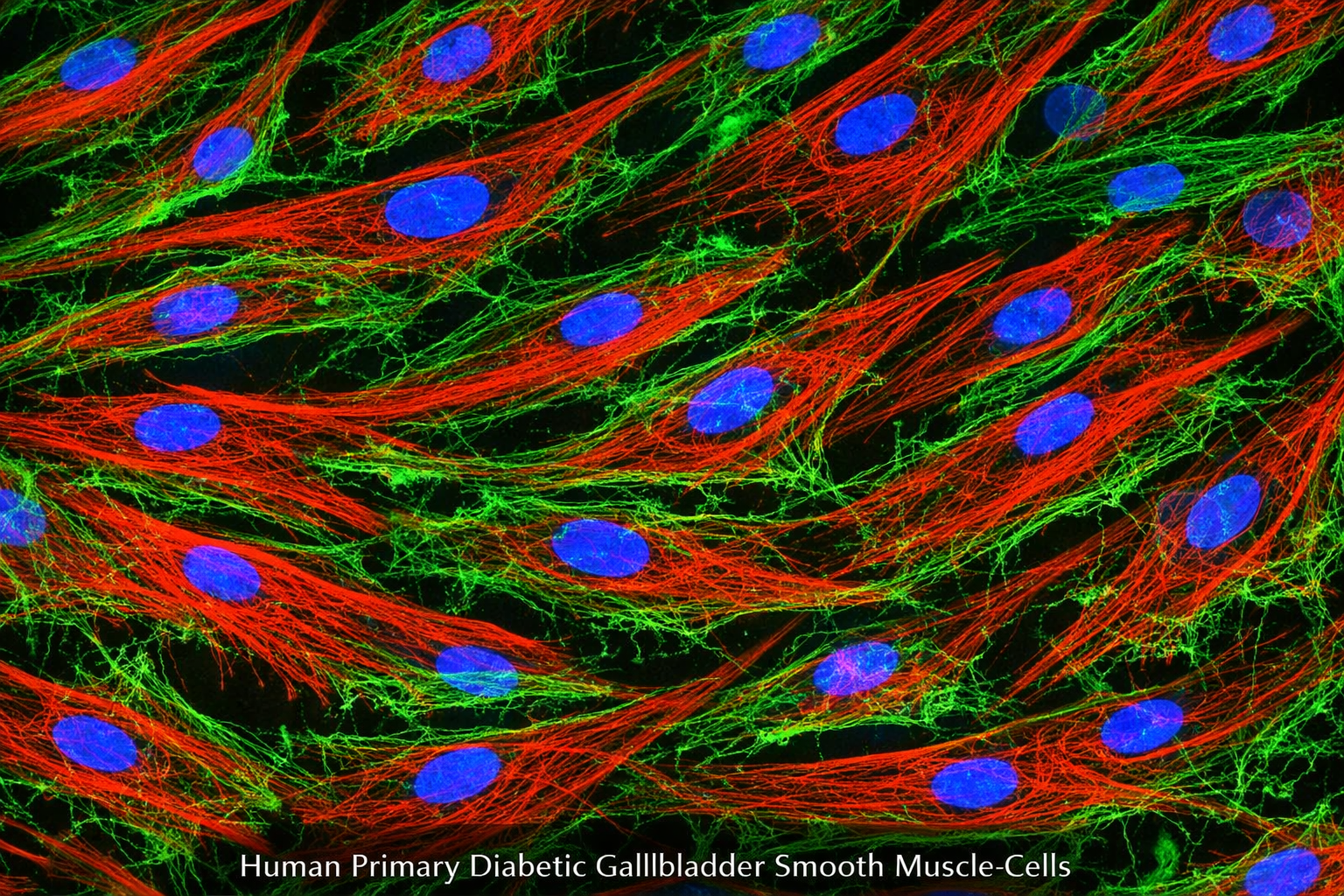
Human Primary Diabetic Kidney Vein Smooth Muscle Cells

Quick Actions
Product Data Sheet
Technical Documentation
Download detailed specifications, protocols, and safety information
File size: 173.8 KB
Product Overview
Description
Human Primary Smooth Muscle Cells obtained from the renal veins of patients with diabetes, emphasizing their origin, biological properties, and potential role in the pathology of diabetic kidney disease.
Guarantee expansion for a minimum of 2-5 population doublings.
IMPORTANT: Quality warranty requires use of NeoBioPharma media. Biohazardous material despite negative pathogen testing. Quality claims must be reported within 1 month.
Key Features
Catalog Number: NBPDS006
Product Format: Frozen
Vial Cell Number: > 5x10[5] cells/vial
Passage:1(P1)
Storage: Liquid Nitrogen, Vapor Phase
Intended Use: Research use only (RUO)
Characterization Markers
Characterized by positive expression of smooth muscle cell-specific alpha-actin .
Quick Facts
Product Type
Human Type II Diabetes Endothelial Cells
Viability
>95% viable (Trypan Blue exclusion)
Applications
Research & Development
Availability
In Stock • Global Shipping
Technical Specifications
Applications & Use Cases
Research Areas
In Vitro Studies
Cell culture and screening applications
Molecular Biology
Gene expression & signaling studies
Drug Discovery
Screening & validation studies
Disease Modeling
Pathophysiology research
Support & Resources
Technical Support
Our experts are available for technical questions and protocol assistance.
Contact Support